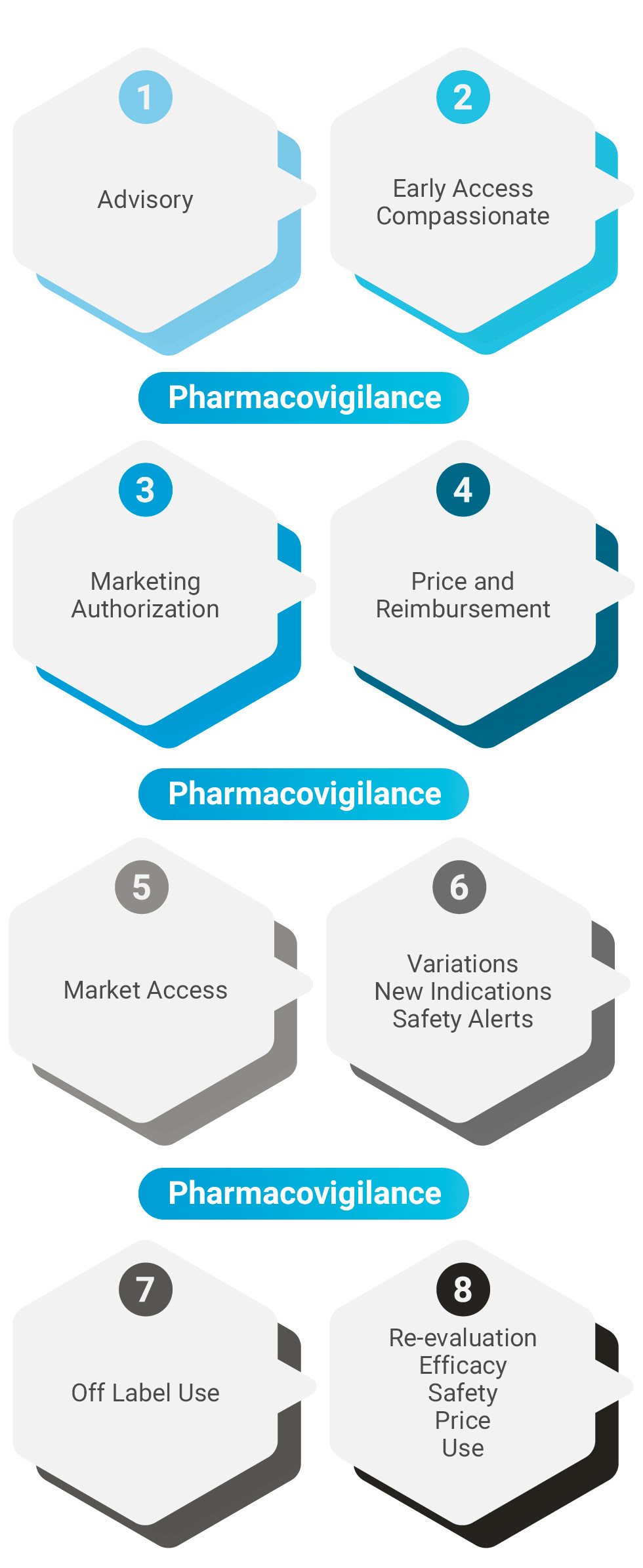
Farmalabel23 offers a comprehensive service to the pharmaceutical industry, starting from the initial phases before the marketing authorization, in the authorization, continuing with the decision on price & reimbursement, and its subsequent incorporation into the market.
Once the product is marketed, a license maintenance service is offered (variations, renewals …), advice on price reviews that may occur due to changes in indications or indicated by the health authorities, and other services required by regulatory agencies.
With regard to pharmacovigilance, Farmalabel23 offers this service to companies that market drugs in Spain, either as a local responsible person or as a EU QPPV.